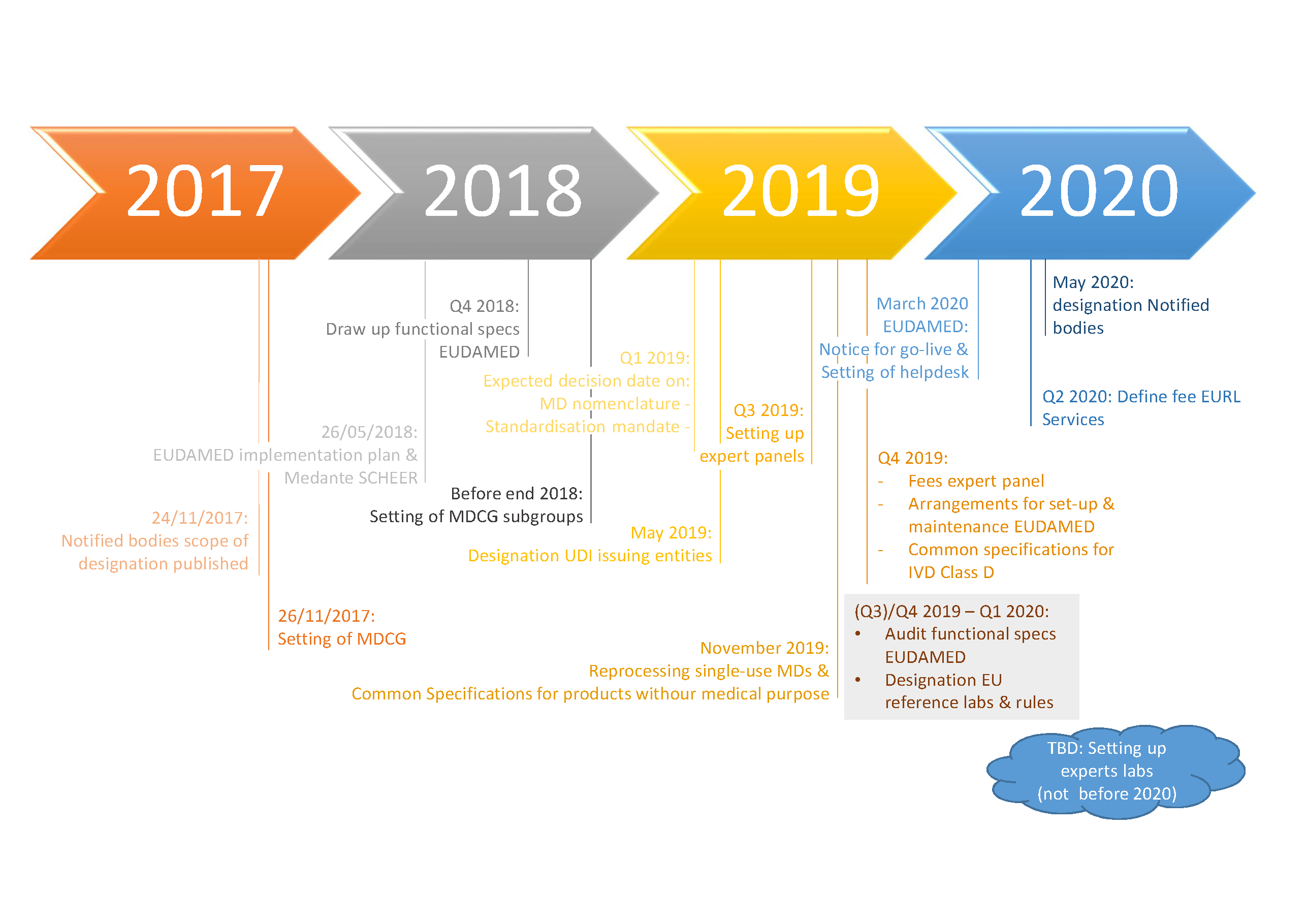
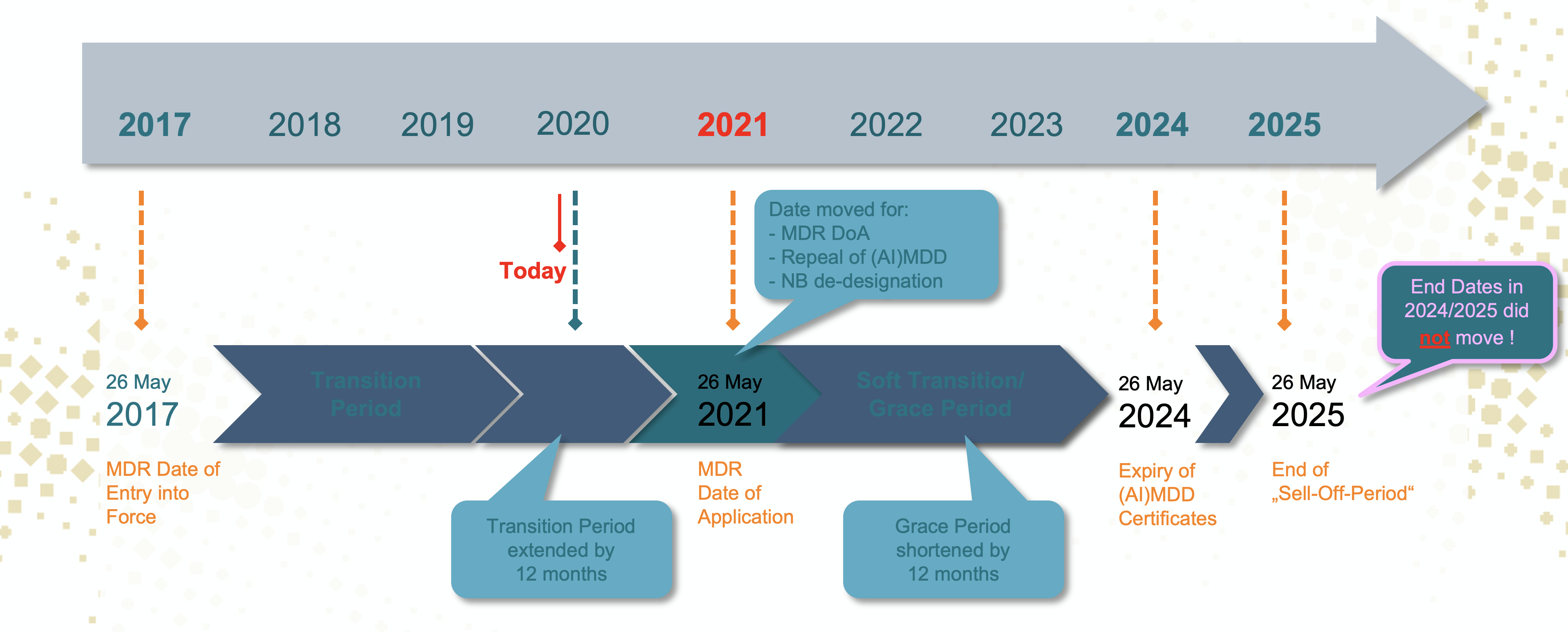
Updated EC Implementation Rolling Plan (MDR/IVDR) · MDlaw – Information platform on European medical device regulations

All Guns Blazing: Commission Brings Sector Up To Speed With Its MDR/IVDR Implementation Plans :: Medtech Insight

Sustainability | Free Full-Text | Complexity Stage Model of the Medical Device Development Based on Economic Evaluation—MedDee